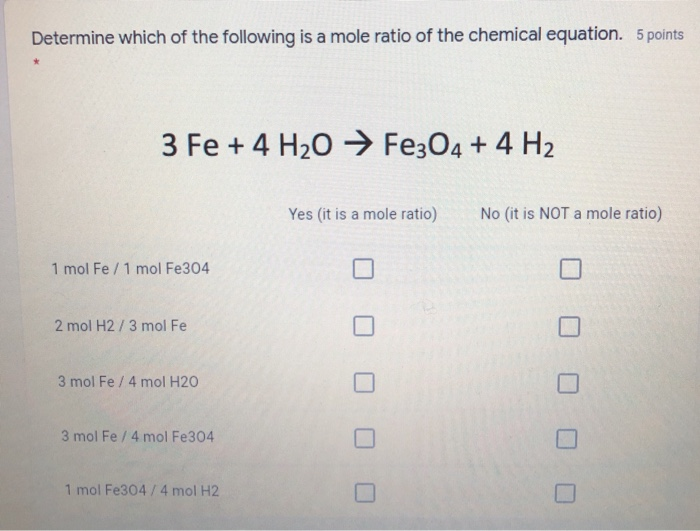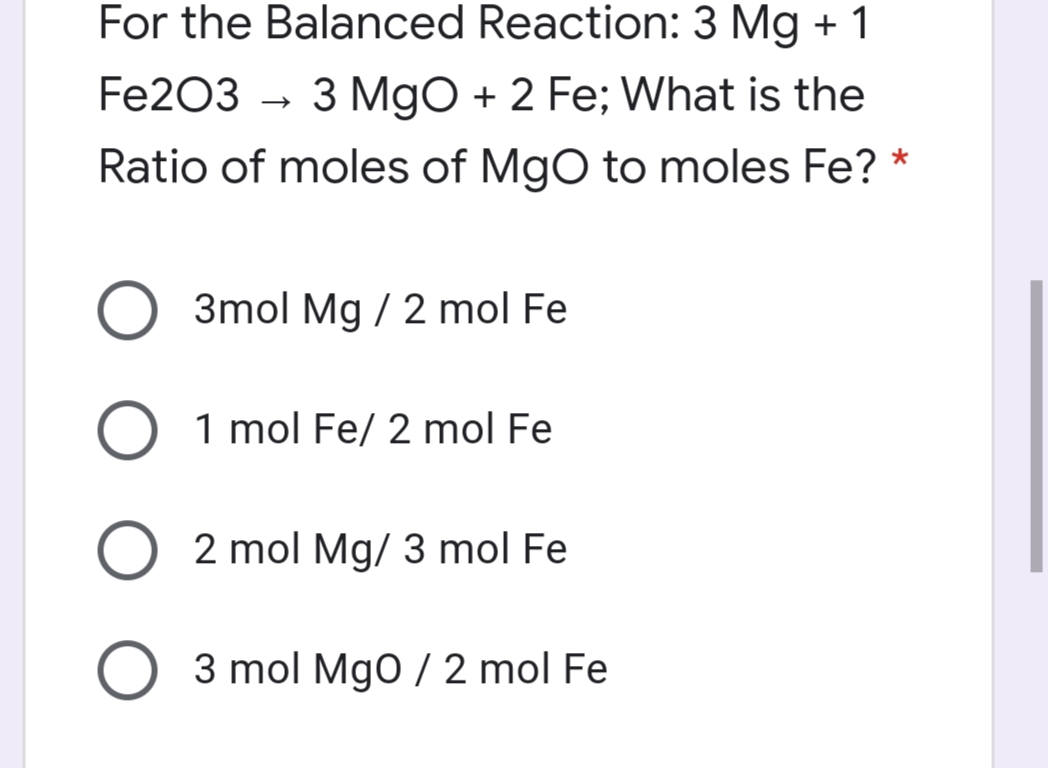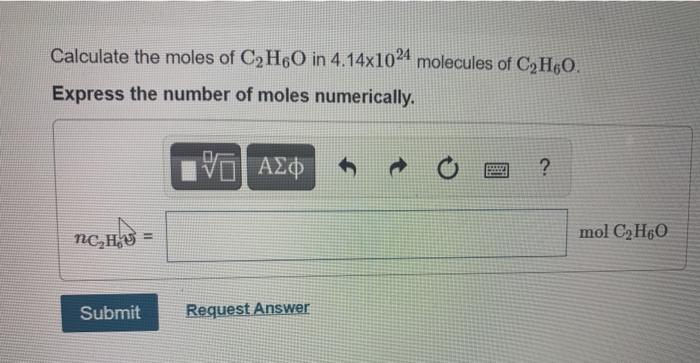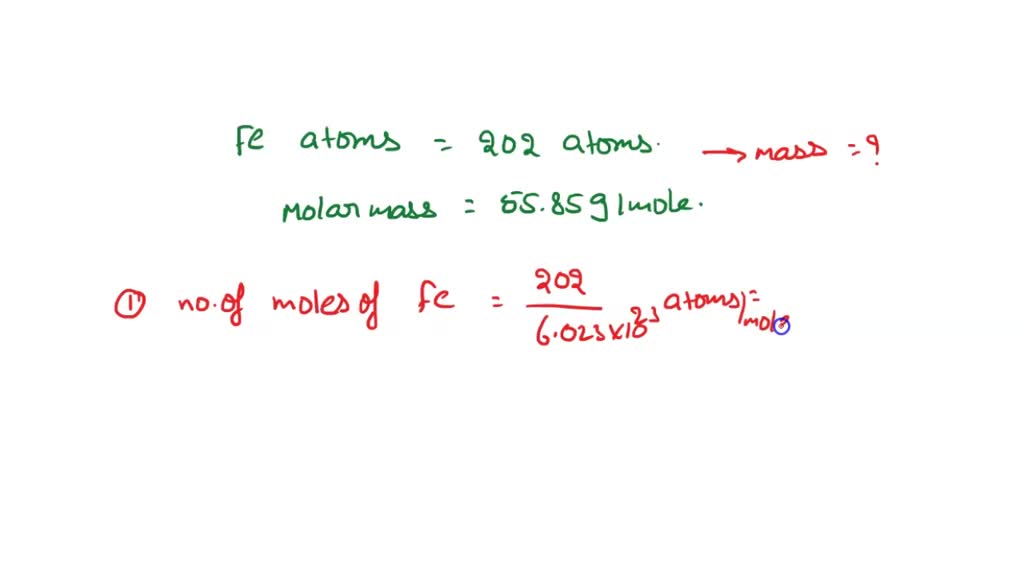
SOLVED: Question 3 of 97 Submit Calculate the mass, in grams, of 202 atoms of iron, Fe (1 mol of Fe has a mass of 55.85 g). 2 3 5 6 8 X10O

Jericho Rayel Timbol on Twitter: "Now let us to moles to mass ratio. Consider the reaction A → B Mass A can be converted to Mass B given that the molar masses
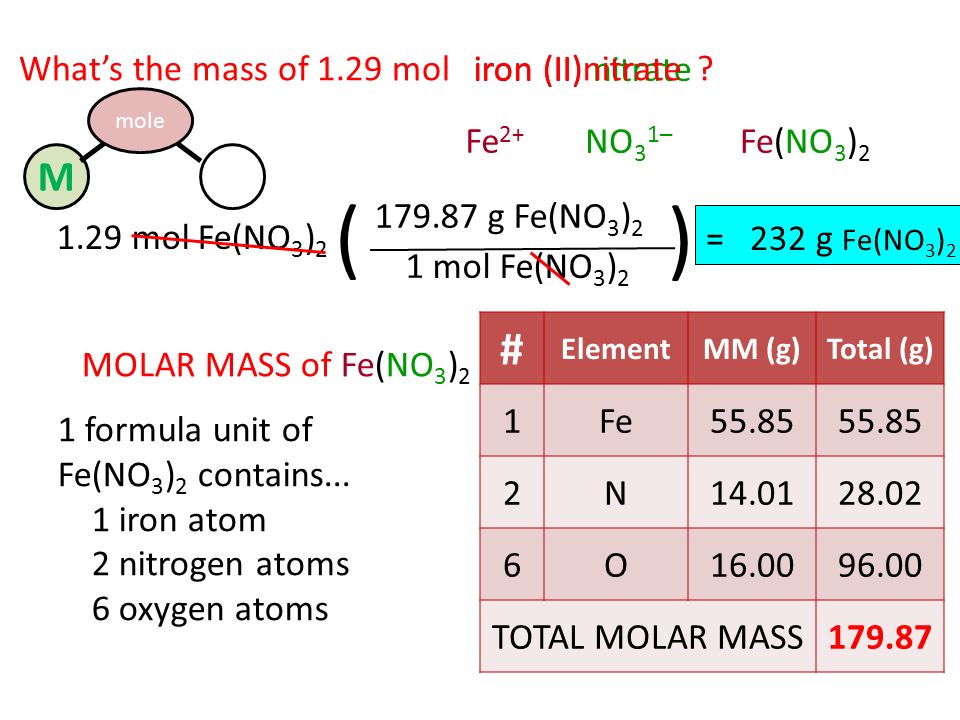
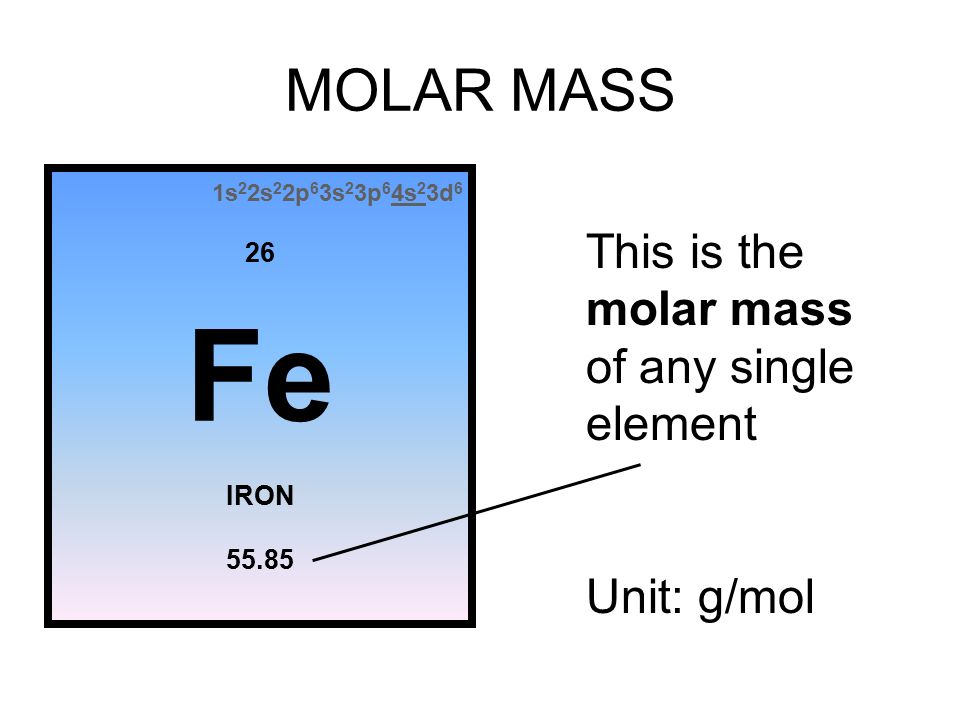
MOLE (mol) Mass (g) Particles (atoms,m'c or f.u.) Which conversion factors that allow you to convert from mass to moles and from particles to moles. - ppt download

Iron standard solution traceable to SRM from NIST Fe(NO₃)₃ in HNO₃ 0.5 mol/l 1000 mg/l Fe Certipur® | Sigma-Aldrich

1 You need 25 mol of iron Fe for an experiment How many grams of iron must you measure out? 2H - YouTube
Bling Bling: if I were to give each of you one atom of gold for every second that has elapsed since the Dinosaur's went extinct 65 million years ago, how. - ppt download
a) Molar magnetic susceptibility of Fe 3 Sb 4 O 6 F 6 (per one mol Fe... | Download Scientific Diagram