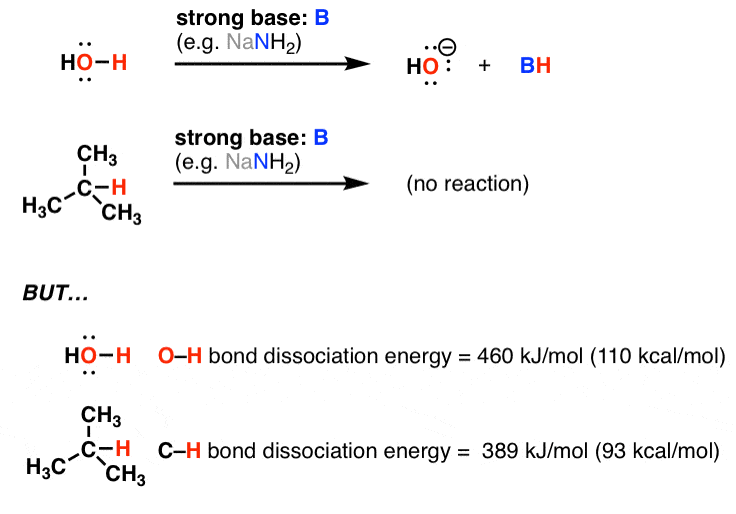
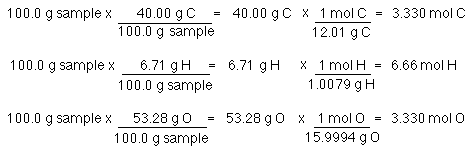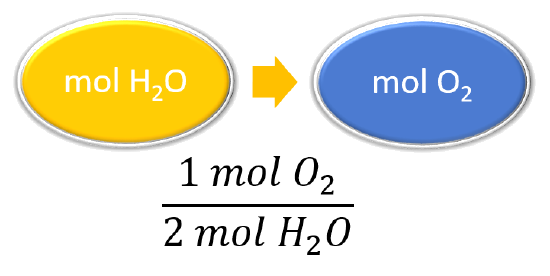Objective: Define empirical formula, and explain how the term applies to ionic and molecular compounds

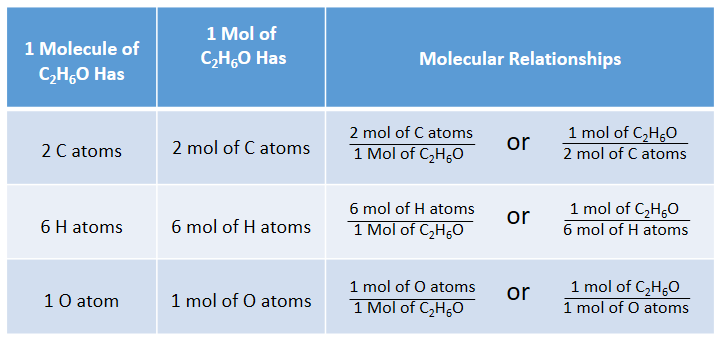
Empirical and molecular formulas for compounds that contain only carbon and hydrogen (C a H b ) or carbon, hydrogen, and oxygen (C a H b O c ) can be determined with a process called combustion analysis. The steps for this procedure are

Bisphenol A, molecular formula: C 15 H 16 O 2, molar mass is 228.29 g/mol. | Download Scientific Diagram

Chemistry Warm Up: Mole / Mass / Particles 1.What is the mass of one mole of water? 2.If one milliliter of water has a mass of 1.00grams, how many moles. - ppt download

Question Video: Calculating the Mass of Solute Needed to Prepare a Solution with a Desired Concentration and Volume | Nagwa

3.16 | Determine the number of moles of compound and the number of moles of each type of atom in - YouTube

Mole Concept and Chemical Calculations: Difference between Relative Atomic Mass, Relative Molecular Mass, Relative Formula Mass and Molar Mass

SOLVED: Determine the number of moles of oxygen atoms in each sample. a. 4.88 mol H2O2 b. 2.15 mol N2O c. 0.0237 mol H2CO3 d. 24.1 mol CO2
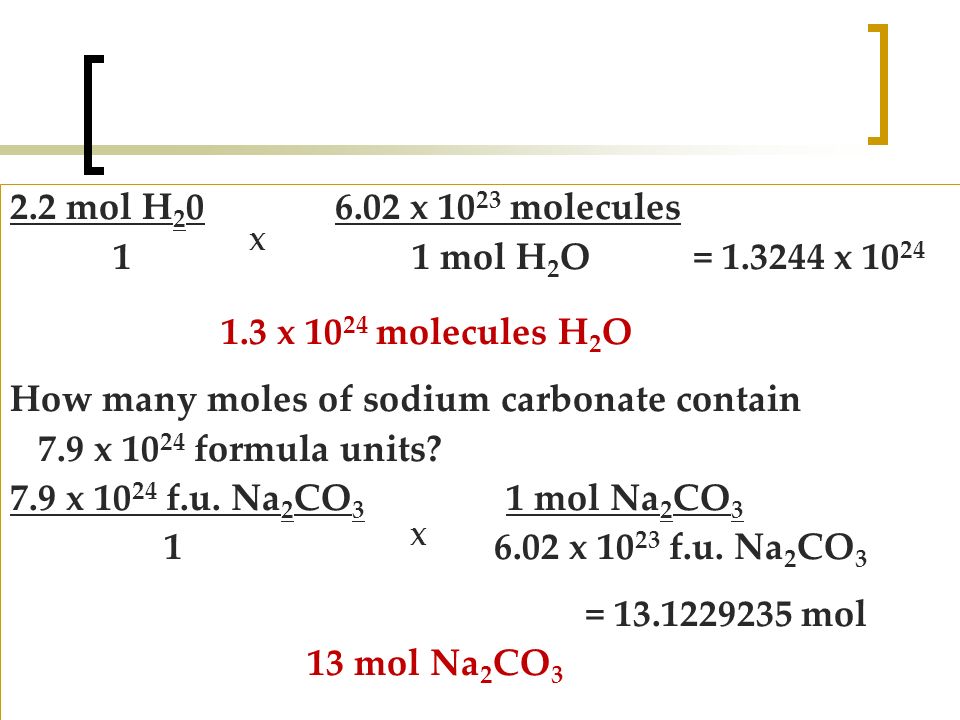
The Mole & Chemical Quantities. The Mole Mole-the number of particles equal to the number of atoms in exactly 12.0 grams of carbon mol = 6.02 x. - ppt download
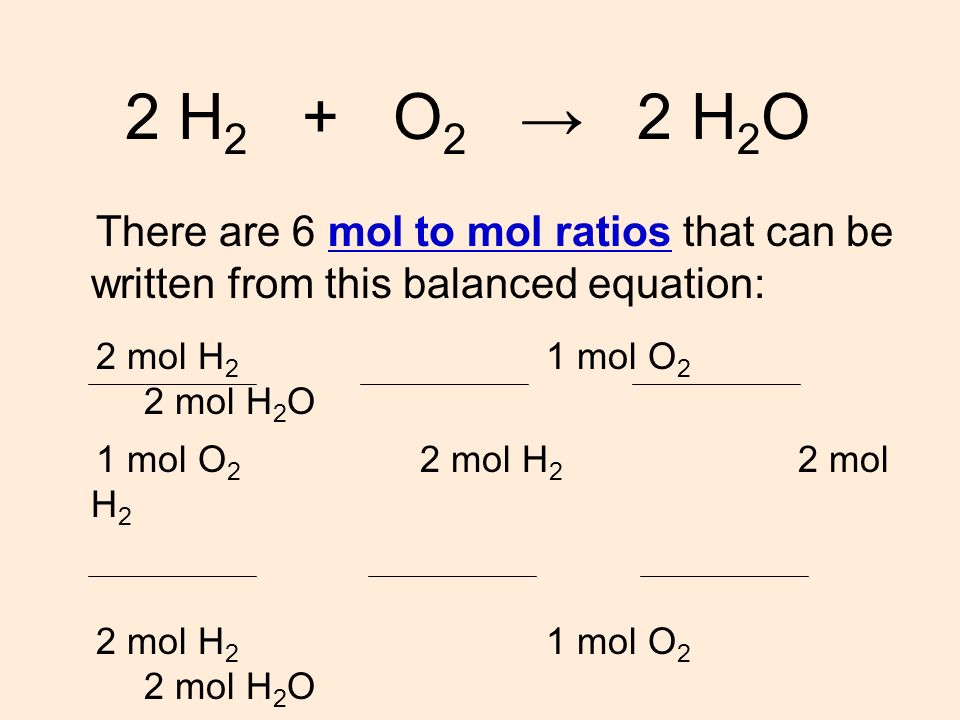
Mol ratio: coefficients of a balanced equation 2 H 2 + O 2 → 2 H 2 O 2 mol H 2 for every 1 mol O 2 In chemical calculations, mol ratios convert moles of. - ppt download